Biological evaluation of medical devices Part 14. Learn more about devices such as diagnostic tests ventilators and personal protective equipment PPEincluding surgical masks face shields.

Market Access For Medical Software In The United States Vde Medical Devices And Software
Recognized Consensus Standards.
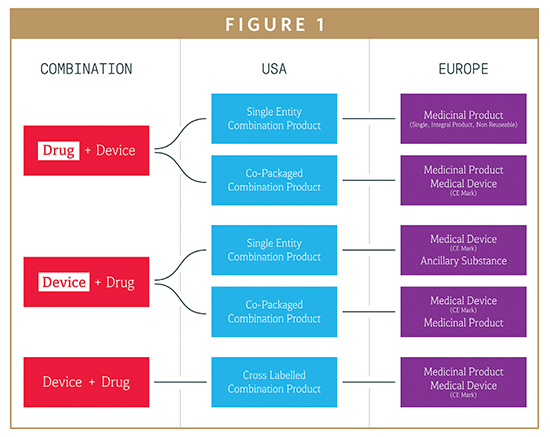
Fda list of recognized standards for medical devices. 21 This Standard is applicable to INTEROPERABLE MEDICAL PRODUCTS including assembled systems of INTEROPERABLE MEDICAL PRODUCTS that comprise or are intended to be incorporated into INTEROPERABLE MEDICAL SYSTEMS within an INTEROPERABLE ENVIRONMENT. To apply the voluntary consensus standards a medical device manufacturer must perform the following steps. It also lists symbols that satisfy the requirements of this document.
Manufacturers should familiarize themselves with the revised FDA list in order to issue accurate Declarations of Conformity and ensure compliance with these standards as part of their US medical device registration efforts. Informational US FDA Modifications to the List of Recognized Standards Recognition List Number. 13 Zeilen Recognized Consensus Standards.
In late October 2019 the US. ISO 14708-32017 is applicable to. After FDA has decided to recognize a standard we will update our online database to reflect the decision even before formal recognition of the standard occurs by publication in the Federal.
Food and Drug Administration FDA published more than 80 additions withdrawals or modifications to its list of recognized consensus standards. FR Notice List 53 Docket No. List of Recognized Standards 12 ISO 141172012Ed10 Active implantable medical devices Electromagnetic compatibility EMC test protocols for implantable cardiac pacemakers implantable cardioverter defibrillators and cardiac resynchronization devices.
The final rule permits the use of symbols in all medical device labeling without adjacent explanatory text referred to as stand-alone symbols if certain requirements are met. Appropriate Use of Voluntary Consensus Standards in Premarket Submissions for Medical Devices FDA. The US Food and Drug Administration FDA has announced changes to the list of recognized standards the agency uses in the medical device regulatory process.
Informational USFDA FDA Modernization Act of 1997. This document identifies requirements for symbols used in medical device labelling that convey information on the safe and effective use of medical devices. Identification and quantification of degradation products.
Food and Drug Administration FDA has published an updated list of consensus standards the agency recognizes as part of its premarket review of medical devices. 102 Zeilen Standard Guide for Assessing Medical Device Cytocompatibility with Delivered Cellular. Identification and quantification of degradation products from polymeric medical devices.
Check the recognition of the standard by the FDA. FDA Modifies List of Recognized Standards for Medical Devices. Medical Device and FDA Regulations and Standards News.
Biological evaluation of medical devices Part 13. Recognized Consensus Standards. FDA Updates List of Recognized Standards for Medical Devices.
These standards are also listed in the Recognized Consensus Standards online database. FR Notice List 52 Docket No. The final rule.
Coronavirus COVID-19 and Medical Devices. Food and Drug Administration FDA has updated its list of recognized international and national standards that can be used to demonstrate compliance with certain requirements for premarket review and authorization of medical devices. The information provided in the database includes both the recognition.
All consensus standards recognized by the FDA have a recognition number assigned in a special notice published in the Federal Register. Modifications to the List of Recognized Standards Recognition List Number. They can be found below.
22 This Standard specifies a baseline set of requirements for. The new lists of references of harmonised standards for medical devices have been published OJ L 0901 of 25 May 2020. New 2020 lists of harmonised standards for medical devices are now available.
FR Notice List 51 Docket No.

Medical Device Standards State Of The Art By Sam Lazzara
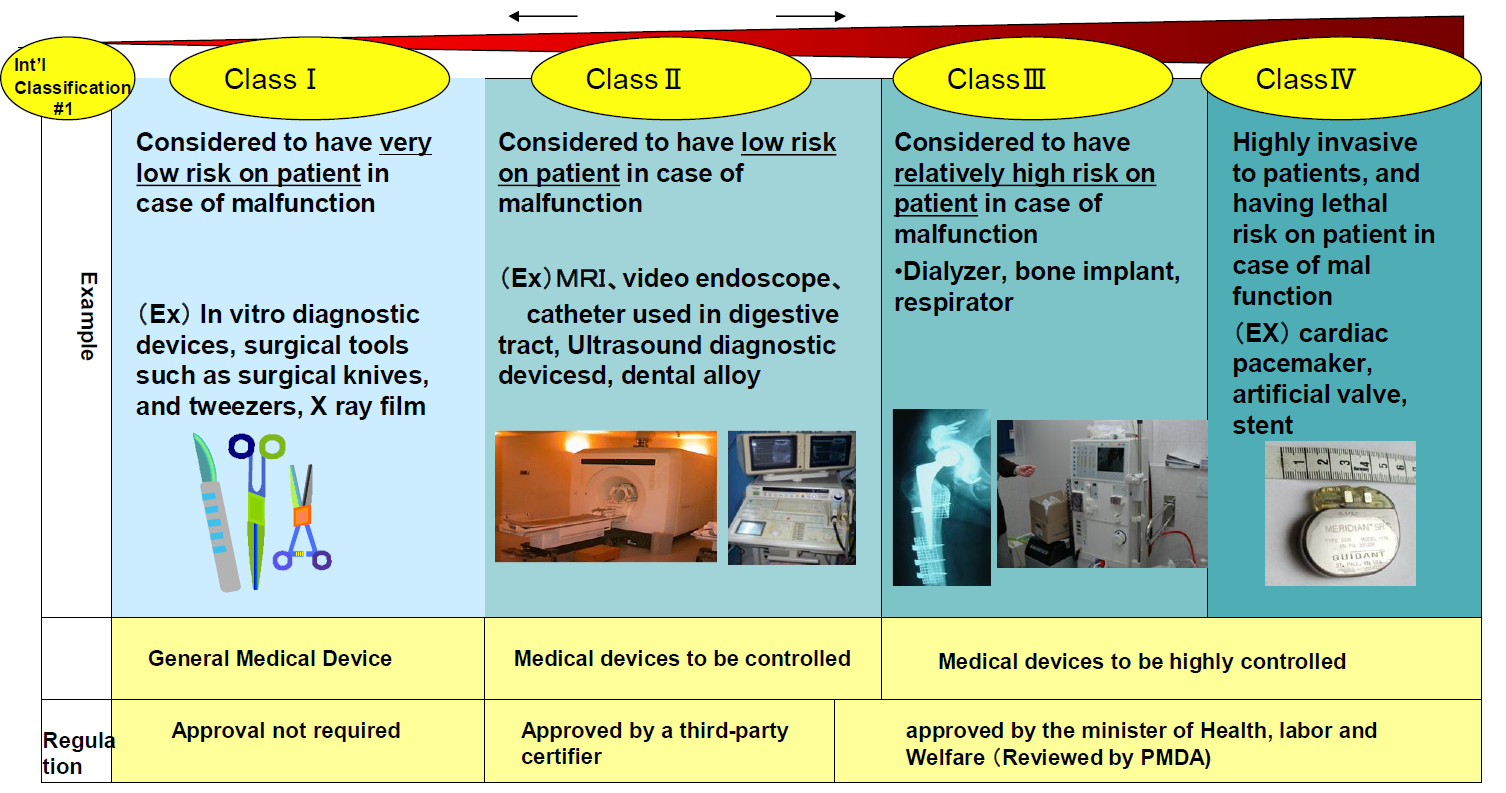
Interoperability Standards For Medical Device Integration In The Or And Issues Relating To International Approval Procedures Part 4 Iscasblog

How Cybersecurity Requirements Will Engage Medical Device Manufacturers In The Future

Device Regulations The New Medical Device Regulation The Applicability Of Article 117 To Medicinal Products
