Food and Drug Administration FDA issues recalls and safety alerts to keep consumers and the healthcare community informed. 34 Zeilen Abbott Vascular Recalls NC Trek RX and NC Traveler RX Coronary Dilatation Catheters.

Software As Medical Device Samd Classification And Definitions
Actions for Clinical Users.

Fda medical device alerts. FDA regulates the sale of medical device products in the US. Sent a New Urgent Medical Device Recall letter to all affected customers and provided the following instructions. List of field safety notices FSNs from medical device manufacturers from 14 December to 18 December 2020 Alert type.
In September the FDA issued multiple new safety alerts for medical devices and drugs. 04202021 Detention Without Physical Examination of Devices without Approved PMAs or IDEs and Other Devices Not Substantially Equivalent or Without a 510k. On March 3 2021 CareFusion 303 Inc.
And monitors the safety of all regulated medical products. Please note that the FDA lists medical device recall notices by the date that it posts the recall rather than the recall initiation date. FDA can spot safety signals in Medical Device Reports MDRs submitted to its Manufacturer and User Facility Device Experience MAUDE database medical.
Field safety notice Issued. 03172021 Detention Without Physical Examination of Devices from Firms that Have not met Device Quality System Requirements 89-08. In Medical Device Recall Posted October 17 2019 The US.
The FDA monitors reports of adverse events and other problems with medical devices and alerts health professionals and the public when needed to ensure proper use of devices and the health and safety of patients. Consumers can find information about how such devices are registered their product life cycle and recalls. MedWatch - your FDA gateway for clinically important safety information and reporting serious problems with human medical products Safety Alerts for Human Medical Products Drugs Biologics.
On The Go. The FDA posts Medical Device Safety Communications to describe FDAs analysis of a current issue and provide specific regulatory approaches and clinical recommendations for patient management. Affordable BBB backed mobile medical alert systems built by an FDA registered manufacturer.
Safety alerts often involve drugs medical devices supplements and cosmetics. Browse by Product Area. Mobile Help Wired Home.
You can find the date that a firm initiated a recall in. This database contains Medical Device Recalls classified since November 2002. Import Alert Type Publish Date Import Alert Name.
The FDA monitors reports of adverse events and other problems with medical devices and alerts health professionals and the public when needed to ensure proper use of devices and the health and. The Center for Devices and Radiological Health CDRH is an FDA component and looks after this program. Import alerts inform the FDAs field staff and the public that the agency has enough evidence to allow for Detention Without Physical Examination DWPE of products that appear to be in violation.
Since January 2017 it may also include correction or removal actions initiated by a. MobileHelp medical alert systems keep seniors safe at home and while away. As such the FDA maintains multiple medical device databases several of which should be of interest to people researching medical alert providers.
Medical Devices Alerts MDAs are the prime means of communicating safety information to HSC organisations and the wider healthcare environment on medical devices.
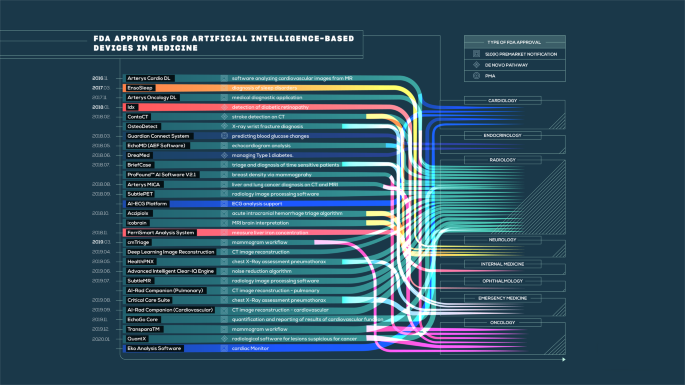
The State Of Artificial Intelligence Based Fda Approved Medical Devices And Algorithms An Online Database Npj Digital Medicine

Fda Issues Multiple Safety Alerts For Medical Devices Drug And Device Watch

Fda Issues Draft Guidance For Testing And Labeling Medical Devices For Mri Knowledge Exponent

Fda Warning Letters Over Medical Devices 2009 2017 Statista
