Batch Release certificate is a documented evidence of quality testing carried out by the manufacturer of Medical Devices in batch ways. Our comprehensive testing methods ensure safe and effective performance of a wide variety of medical devices and medical products.
As an independent lab we also provide validated testing.

Medical device lot release testing. Guidance regarding the fabrication packaginglabelling testing distribution and importation of medical gases is provided in the Good Manufacturing Practices for Medical Gases GUI-0031. Batch Release certificate play many role and GMP standard require that manufacturer should test against the specification for. Medical device lot release testing ensures that your complex catheter designs get to market safely.
The content of this document should not be regarded as the only interpretation of the GMP Regulations nor does it intend to cover every conceivable case. Generally 10 to 20 spore strips are used to monitor a cycle. However end product sterility testing of medical devices is occasionally performed as part of investigations or to support other information in making a lot release decision.
Bulk Lot Release Testing. Testing for medical devices should be conducted using rinsingeluting and sampling techniques as described in ISO 10993-1 14 and ISO 10993-12 15 as also used for inhibitionenhancement. Final Product Release Testing.
Special processes are processes for which the output cannot be verified fully verified per. The need to provide adequate and reliable sterility test data. An understanding of sterility testing is beneficial in terms of designing a validation process.
18 Note 2 Quality control may monitor all or part of the measurement procedure from 19 the collection of samples to reporting the result of the measurement. In short Batch Release certificate is a piece of paper that certifies analysis. This test is appropriate for devices sterilized by steam or ETO in a validated cycle.
These teams include chemists material specialists toxicologists and biocompatibility testing experts. Lot release testing is an integral part of quality control as well as sterilization confirmation. A medical device whether used.
We perform product-specific testing protocols that are conducted prior to the final release of the product. NAMSAs medical device testing team is comprised of various experts readily available to evaluate your device. SGS PSI medical device testing services include.
The selected tests include but are not limited to tests to ensure that special process outputs are verified. Lot release is a mechanism that provides FDA with a real-time system to continuously monitor product quality through review and testing of many of the biological products that it regulates. Instead of testing quality coming out of the process at every step from raw materials to finished goods with a complete self-auditing eDHReBR.
USP requirements employ sterility testing as an official test to determine suitability of a lot. It can provide an accurate basis for calculating effective sterilisation doses for a given therapeutic and will provide the quantity of viable microorganisms in or on a therapeutic medical device or raw material before sterilisation. The lot release testing I have seen typically involves a statistical sampling of devices after sterilization if applicable that are subjected to performance tests that are selected based on risk.
Siemens Medical Device and Diagnostics solution has been deployed with impres- sive results in a variety of medical device product segments including diagnos-tics orthopedics cardiovascular hospital and wound care diabetes care renal blood. Whats lot release testing batch release testing. 16 Note 1 In the IVD medical device industry and in many laboratories that use IVD 17 medical devices these activities are commonly referred to as quality control.
Pharmaceutical Release Testing To support your commercial product and clinical trial material release testing needs Eurofins BioPharma Product Testing offers the capacity and breadth of capabilities to test your drug substance drug product or in-process materials in a timely manner. Lot release testing tests a batch of drug substance or finished product against the registered specification for that material to ensure compliance for all attributes. Bioburden testing is applicable for.
Sterility testing of pharmaceutical articles is required during the sterilization validation process as well as for routine release testing. Together we can ensure that your safety evaluation plan minimizes unnecessary testing maintains compliance with all pertinent standards and regulations and provides evidence for. Spore strips are cultured in SCDM and then usually incubated for 7.
4 20 In vitro diagnostic medical device IVD. We provide batch and lot release testing for active pharmaceutical ingredients APIs investigational medicinal products IMPs and finished products with Marketing Authorisation across a wide range of pharmaceutical products including or inhaled drug products oligonucleotides and biologics such as monoclonal antibodies biosimilars bispecific antibodies and viral vectors. We are FDA registered cGMP compliant and DEA licensed.

Process Validation Or Verification Medical Device
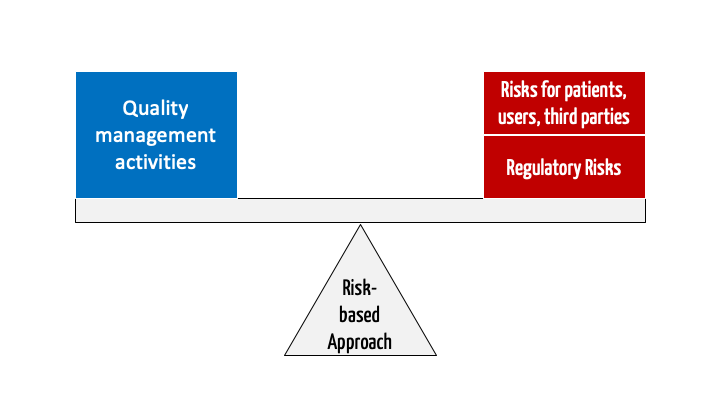
Risk Based Approach How To Fulfill The Iso 13485 Requirement

Software As Medical Device Samd Classification And Definitions

Technical Documentation Technical File Precondition For Approvals

