The biocompatibility evaluation of a product is not intended to be taken lightly or just some regulatory hurdle you must jump. Issued an urgent safety advisory over four patient deaths related to its HeartMate II left ventricular assist device LVAD.

Astonishing Medical Supplies Dog Lovers Medicalassistant Medicalequipmentplanes Biomedical Engineering Engineering Design Data Science
No products need to be returned because the deaths and serious injuries were not the result of device failure but rather.

Failed biomedical devices. 1 The X-ray machine images internal organs and thus discovers internal abnormalities and tumors in time to remove them. The development of biomedical devices requires that the above considerations are not just. In any case the test device failed the test.
Electronic photonic and MEMS-based devices. When failures occur the company selling the device will often have to step in and publicly recall all affected copies or at the very least issue advice to customers on the problem. Besides diagnosis and treatment of diseases the biomedical engineering also plays a big role towards to alleviating rehabilitation and compensating for disabilities andor injuries.
Inadequate instructions component issues and assembly errors--those are but some of the causes behind recent medical device recalls and serious warnings announced by the FDA. In many cases these biomaterials are derived from an individualss own cells. Regenerative medicine CAR-T and gene therapy are three significant areas of biomedical device.
Frequently charging an old phone may be acceptable in the consumer device space but a medical device that supports vital functions will be held to a higher standard. 2 Computed tomography generates slice images of internal organs with improved contrast and spatial resolution. The series prompted the FDA to publish its entire hidden database online revealing 57 million device-related injuries or malfunctions.
This phenomenon is called fatigue failure and responsible for the majority of failures in many mechanical components. The cytotoxicity test is a very sensitive test and is the most likely test to cause you anguish. Here are some of the major medical device failure events since January.
2 After peeling off your PDMS clean the PDMS surface with 3M magic tap. MPMNThis issue can even extend to instructions as demonstrated by two medical device failure issues that recently caught the FDAs eye. This publication was produced by the Department of Essential Health Technologies of the Health Systems and.
To recognize the convergence of bio-engineering and medical devices the term biomedical device is used. Our biomedical white paper series explores topics on. Summaries of information about the most serious medical device recalls.
Medical device sterilization validation studies are performed for all processes including radiation sterilization by either gamma radiation or electron beam ethylene oxide gas sterilization hydrogen peroxide sterilization and thermal sterilization both moist and dry heat. First and most important thing to do is take a deep breath. Recent Medical Device Recalls Medical device recalls that may potentially present significant risks to consumers or users of the product.
Different from the breakdown in the tension test where the specimen is subjected under gradual increase of loading until fractures failure of a component practically occurs after a lengthy period of repeated stress or strain cycling. The most important biomedical engineering devices are those that save the most lives andor improve the lives of the most people. It is a step-wise evaluation that increases in rigor as the.
3 Magnetic resonance imaging. Inadequate Sterilization for an Orthopedic Surgery Tool. These products are on the list because there is a reasonable chance that they could cause serious health problems or death.
At NAMSA were known for our ability to design custom protocols that not only meet clients testing budgets but. Medical device failures and malfunctions invariably harm peoples wellbeing due to the fact they are often deployed on the frontline of patient care and in users homes. To avoid this kind of failure the stress level.
The critical nature of biomedical device operation demands that functionality and operation be checked and rechecked as failure is not an acceptable option. 1 Before pouring the PDMS onto your mold blow some air on the mold to remove potential unwanted particles. Some of the typical examples of biomedical.
Semiconductor technology is becoming increasingly important in global healthcare enabling novel understanding discovery and treatment of disease to make healthcare more affordable and efficient both in and out of the clinic. The Priority Medical Devices project is the result of a collaboration initiated in 2007 between the Dutch Ministry of Health Welfare and Sport and the World Health Organization. Decreased runtime can also lead to a failure of therapy delivery which can be life-threatening for critical devices.
This type of failure affects the patient on a daily basis and will decrease trust in the medical device and treatment. Medical devices are rapidly advancing from traditional hardware-based systems to include or be biological materials. The aim of the biomedical engineering field is to develop systems equipment and devices in order to solve clinical problems both diagnostics and therapeutics.
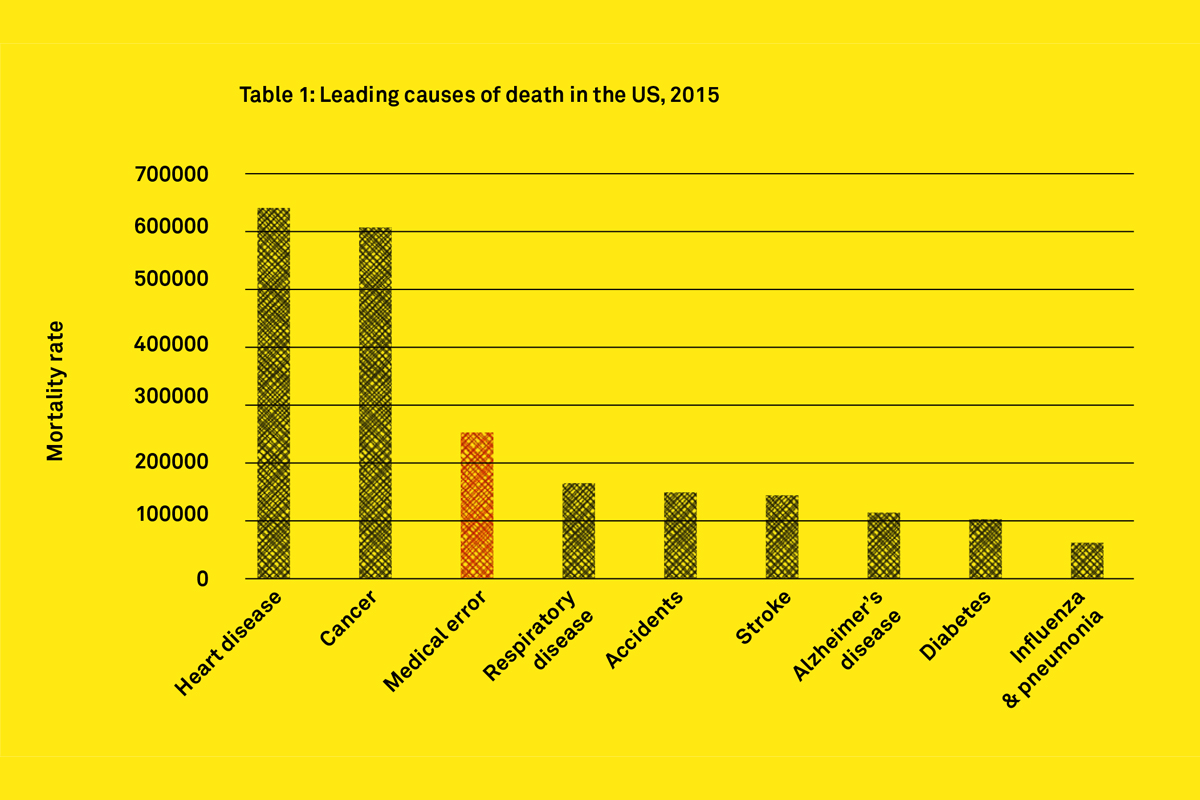
Medical Device Failure An Engineer S View Team Consulting

Spinal Cord Stimulators Market Spinal Cord Stimulator Spinal Cord Spinal

Year 6 Cv Template Cvtemplate Template Biomedical Engineering Engineering Resume Resume Examples

Medical Device Failure An Engineer S View Team Consulting
