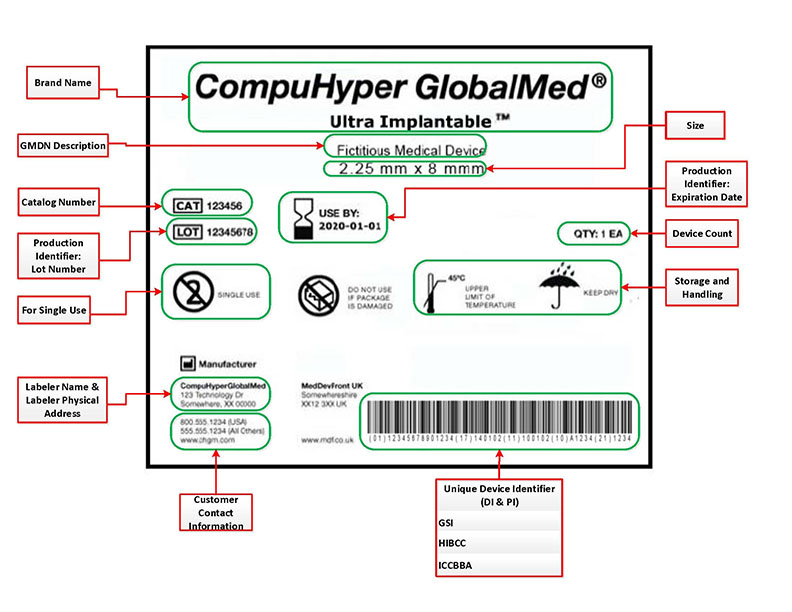
Medical Device Labelling. The guidance is also intended for industry use in preparing device labeling.
Fda Clarifies Udi Form And Presentation In Device Labeling
Later sections in this chapter discuss.
Fda labeling guidance medical device. Information on safety use and performance. 80130 - General exceptions from the requirement for the label of a device to bear a unique device identifier. This guidance document describes the general labelling principles for medical devices and IVD medical devices and supersedes an earlier version produced under the Global Harmonization Task Force GHTF entitled Label and Instructions for Use dated September 16 2011 GHTFSG1N702011.
Medical Device Label and Labeling Introduction to Medical Device Labeling Label vs. 1 to assist manufacturers in their development and 2 to assist Center reviewers in their review and evaluation of medical device patient labeling to help. 8014 emphasis added.
Labeling claims advertising matter or oral or written statements or circumstances that show the medical device is with the knowledge of the manufacturer offered and used for a purpose for which it is neither labeled nor advertised 21 CFR. RAs require and specify information that manufacturers are expected to incorporate in the labelling when the device is placed onto the market. Name and place of business of manufacturer packer or distributor.
The focus of these controls is to ensure that labels are accurately designed and properly applied to the correct products. Food and Drug Administration FDA develops and administers regulations under authority granted by laws passed by Congress that apply to food drugs cosmetics biologics radiation-emitting electronic products and medical devices. Information to be Provided by Manufacturers for the Reprocessing and Sterilization of Reusable Medical Devices 2011-06-01 Private Label Medical Devices.
This guidance is to be used in the preparation of labelling material for non- in vitro diagnostic devices. Labeling regulations pertaining to medical devices are found in the. The intent of this document is to outline the foundational labelling.
This guidance is intended to ensure the adequacy of and consistency in device labeling information. Under the Draft Guidance posted by the FDA the labeling requirements on medical devices which must outline intended use and warnings may soon include a list of 14 network and security-related warnings. Questions and Answers 2011-03-29 Application for a New Medical Device Licence for a Private Label Medical Device 2011-03-29 Guidance Document - Private Label Medical Devices 2011-03-15 Guidance Document.
These regulations specify the minimum requirements for all devices. That makes sense when we consider the trend in the number of connected devices. If your medical device company is planning to sell devices in the United States you will need to comply with the FDA QSR for Labeling and Packaging Control of medical devices found in 21 CFR Part 820120.
13 Scope and Application. Medical devices offered or imported for sale or use in Canada must meet the labelling requirements listed in sections 21 - 23 of the Regulations. It is intended for users of medical devices including IVD medical devices both professional and lay as appropriate and for relevant third parties.
A The label of a device in package form shall specify conspicuously the name and place of business. FDA determines the intended use of a medical device by looking at a wide body of evidence such as. This draft guidance document provides Food and Drug Administrations recommendations on testing to assess the safety and compatibility of medical devices in the Magnetic Resonance MR Environment.
Manufacturers of reusable medical devices are responsible for having labeling that bears adequate directions for use including instructions on preparing a device for use. This includes activities for labelling control incoming control of materials including labelling and design documentation to ensure labels have adequate space on the device. Overview of FDA regulations The FDA requirements for labelling of medical devices are embedded with Quality System Regulation requirements mentioned in 21 CFR Part 820.
At a recent medical device conference 40 of companies pitching investors had a degree of. This guidance provides information for firms about how FDA evaluates firms medical product communications that fall within the scope of FDAs regulatory authority product communications and. This guidance serves a dual purpose.
The general labeling requirements for medical devices are contained in 21 CFR Part 801. 80135 - Voluntary labeling of a device with a unique device identifier.

Fda S Unique Device Identifier Successful Implementation

Fda Issues Final Rule On Symbols In Medical Device Labeling
Fda Unique Device Identifier Rule Starfish Medical