Please note that Class 2 devices and most Class I devices are NOT exempt from GMP requirements. Once you find your device in the panels beginning list go to the section.

Eu Medical Device Classification Form Easy Medical Device Medical Medical Device Infographic
Condoms and pregnancy tests fall into this category.

Fda class 2 medical device list. A listing of Class I and Class II devices exempt from 510 k requirements is available on the Medical Device Exemptions 510 k and GMP Requirements website. External patient support devices such as hospital beds patient hoists walking aids wheelchairs stretchers. Releasable establishment registration and listing information under the Freedom of Information Act is available by.
For example dental floss and bedpans fall into this category. These are our medium to moderate risk devices. The Food and Drug Administration FDA has also published a list of Class 2 special controls devices subject to certain limitations that are now exempt from the Premarket Notification requirements under the Food and Drug Administration Modernization Act of 1997.
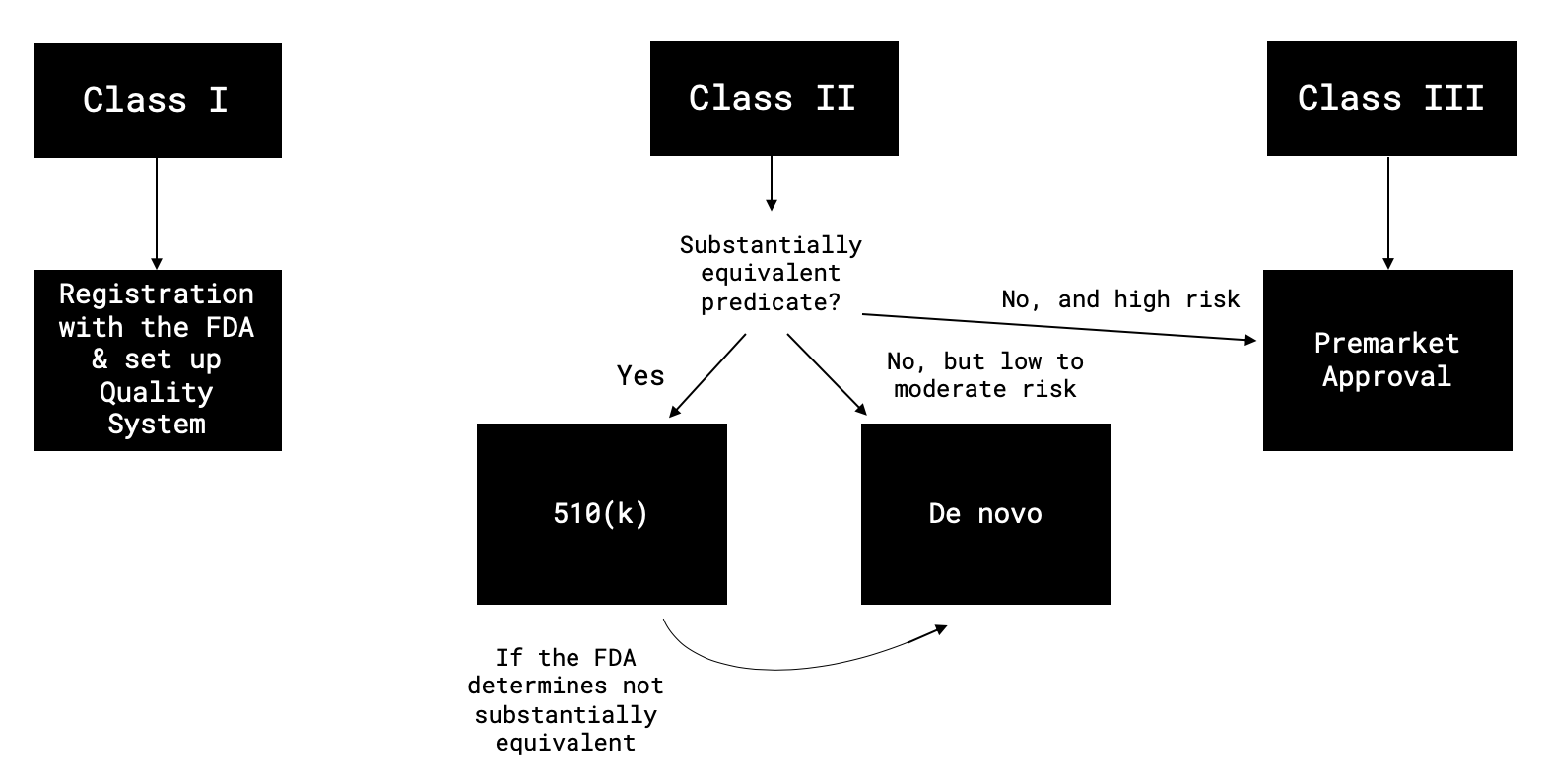
Medtronic Recalls Valiant Navion Thoracic Stent Graft System Due to. There are about 800 Class II medical devices. There are three main classes of medical devices according to risk.
General limitations to the exemptions. As per Rule 2 medical device intended for channeling or storing for eventual administration except with the use of blood and connected to an active device is class 1 medical device. Medical Devices - Class 2 Reorder Description US FDA Code FDA Class Health Canada Code HC Class HC License 1241 Ultrasound Gel 025 liter 85 fl oz Blue - 12Cs MUI 2 80KDQ 1 NA 1243 Ultrasound Gel 13 gal 5 liters Blue - 4Cs MUI 2 80KDQ 1 NA 1245 Ultrasound Gel 025 liter 85 fl oz Clear - 12Cs MUI 2 80KDQ 1 NA.
Class II devices require general controls and special controls. Each classified device has a 7-digit number associated with it eg 21 CFR 8802920 - Clinical Mercury Thermometer. Class 1 These are very low-risk devices and drugs.
The Food and Drug Administration FDA has also published a list of class II special controls devices those devices are annotated as II subject to certain limitations. Establishment Registration and Medical Device Listing Files for Download. As simple as a tongue depressor As complex as robotic surgery devices A medical device.
For example pacemakers and heart valves. List of Medical Devices by Product Code that FDA classifies as Implantable Life-Saving and Life-Sustaining Devices for purposes of Section 614 of FDASIA. Medical device as any product that does not achieve its purposes by chemical action or metabolization.
Class 2 These devices have a higher risk than Class 1. 9 Zeilen Device Name. Class 3 These devices present a very high risk to human health.
Fda Medical Device Regulation 101 By Sindy Shi Hcvc

How The Us Fda Classifies Medical Devices Regulatory Affairs Regulatory Compliance Risk Management

Market Access For Medical Software In The United States Vde Medical Devices And Software
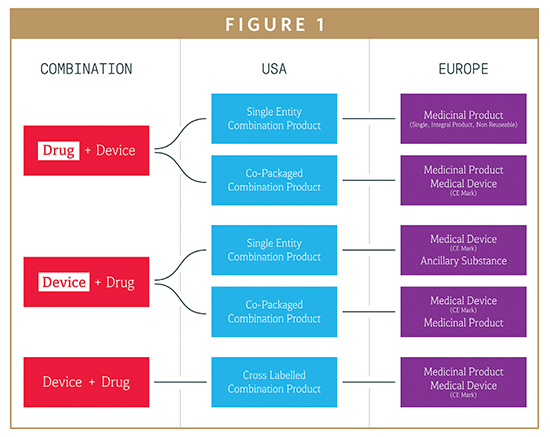
Device Regulations The New Medical Device Regulation The Applicability Of Article 117 To Medicinal Products
