Application Form for Change in Medical Device Registration. Best viewed with 1024x768 pixel or above is recommended.
Taiwan has one of the most enviable healthcare systems in the world it boasts the number one ranking among 93 countries two years in a row from mid-2018 and mid-2020 in terms of the overall quality according to the Health Care Index by Country released by NUMBEO.

Taiwan fda medical device. No161-2 Kunyang St Nangang District Taipei City 11561 Taiwan ROC MAP TEL. The Quality System approval stage and the device. Click here to visit the Taiwan FDA authority website.
The TFDA rests under the authority of The Ministry of Health and Welfare MOHW who performs on-site inspection for local manufacturers and reviews Quality System Documentation QSD provided by foreign manufacturers. Yes medical devices do require registration before being sold in Taiwan. The registration of imported medical device in Taiwan is a two-stage process.
Medical device companies interested in commercializing their products in Taiwan must comply with registration requirements of the Taiwan Food and Drug Administration TFDA. Pre-market approval is necessary for all classes of medical devices prior to Taiwanese market entry. CE certificate if available Requirements for medical device registration in Taiwan.
Best viewed with 1024x768 pixel or above is recommended. Taiwan Technical Guidelines on Medical Device Software. All imported medical devices must obtain a registration certificate from the TFDA.
Taiwans classification system is. Determine classification of your device according to the device database. Under the MOHW the Taiwan Food and Drug Administration TFDA is Taiwans equivalent of the US.
Pre-market approval is required for all classes of medical devices and consists of two stages. Taiwan FDA hosts 2020 APEC Medical Devices Regulatory Science Center of Excellence CoE Workshop Taiwan Food and Drug Administration has become the full member of ICCR Taiwans New Measures for Management of Medical Masks. These reports have generally been required for the first three years that a new device is on market in Taiwan.
The process which is highly regulated by the Taiwan Food and Drug Administration TFDA. Operations Procedure for Applying for Medical Device Certificates. 886-2-2787-8000 Copyright 2018Taiwan FDA.
886-2-2787-8000 Copyright 2018Taiwan FDA. The 1st stage is applying for QSD license for each manufacturer and the 2nd stage is applying for foreign medical device registration. No161-2 Kunyang St Nangang District Taipei City 11561 Taiwan ROC MAP TEL.
No161-2 Kunyang St Nangang District Taipei City 11561 Taiwan ROC MAP TEL. Appoint a Taiwan Agent to manage your device registration and interact with the. The Medical Devices Act link in Chinese establishes a new medical device regulatory framework in Taiwan via a phased implementation schedule.
The first guidance Medical Device Safety Surveillance Management Measures links in Chinese defines the requirements surrounding submission to the TFDA of Periodic Safety Update Reports PSURs. In Taiwan Medical Devices are regulated by the Taiwan Food and Drug Administration TFDA Taiwans equivalent of the United States FDA. The Taiwan TFDA medical device approval process explained Step 1.
Best viewed with 1024x768 pixel or above is recommended. Application form MAHs company seal and representatives chop is required 2. Taiwan FDA Unique Device Identifier UDI Draft Regulation 08 March 2021 Jenny Lin Taiwan has recently published a draft regulation of their Unique Device Identifier UDI Requirements for Medical Device Labels on 5 November 2020.
A copy of pharmaceutical firm permit license as a. The new approach is based on the regulations adopted in the United States Japan. Some key elements of the new regulation to be enforced by the Taiwan Food and Drug Administration.
In Taiwan are medical devices required to be registered before they can be sold. Application Form for Registering Manufactured Imported or Export-only Class 2 and Class 3 Medical Devices. The Taiwanese government plans to launch new medical device regulations on May 1 2021 ushering in new requirements for unique device identification UDI distribution and other processes.
Requirements for QSD Good Manufacturing Practice. This regulation will be applicable to all imported and local Class II and Class III medical devices. Upcoming update to the Taiwan FDA Medical Devices Act.
Data Sheet and Checklist for DomesticImported Class II and III Medical Devices. 886-2-2787-8000 Copyright 2018Taiwan FDA. The Taiwan Food and Drug Administration FDA has published technical guidelines for medical device software inspection and registration of Artificial Intelligence and Machine Learning technology.
Taiwan Food and Drug Administration TFDA.

Taiwan Medical Device Registration And Approval Chart Emergo

Taiwan Medical Device Registration And Approval Chart Emergo

Taiwan Fda Advances First Medical Device Act 2018 01 16 Fdanews
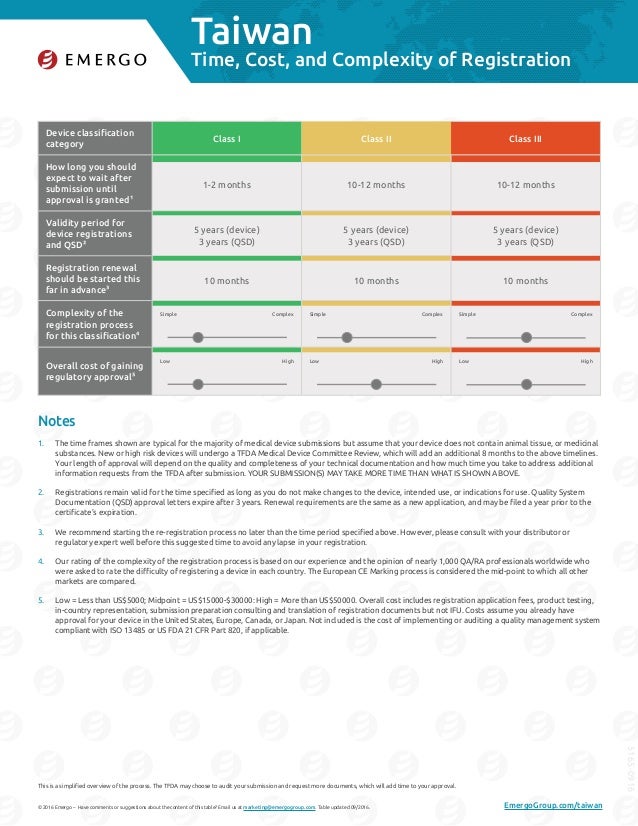
Taiwan Medical Device Registration And Approval Chart Emergo

