The finished design output is the basis for the device master record. Investors and non-investors alike developed some understanding of that FDA timeline.
Https Www Fda Gov Media 131268 Download
Medical device clearance is less understood yet critical for investors looking to benefit from breakthroughs and disruptive medical equipment.
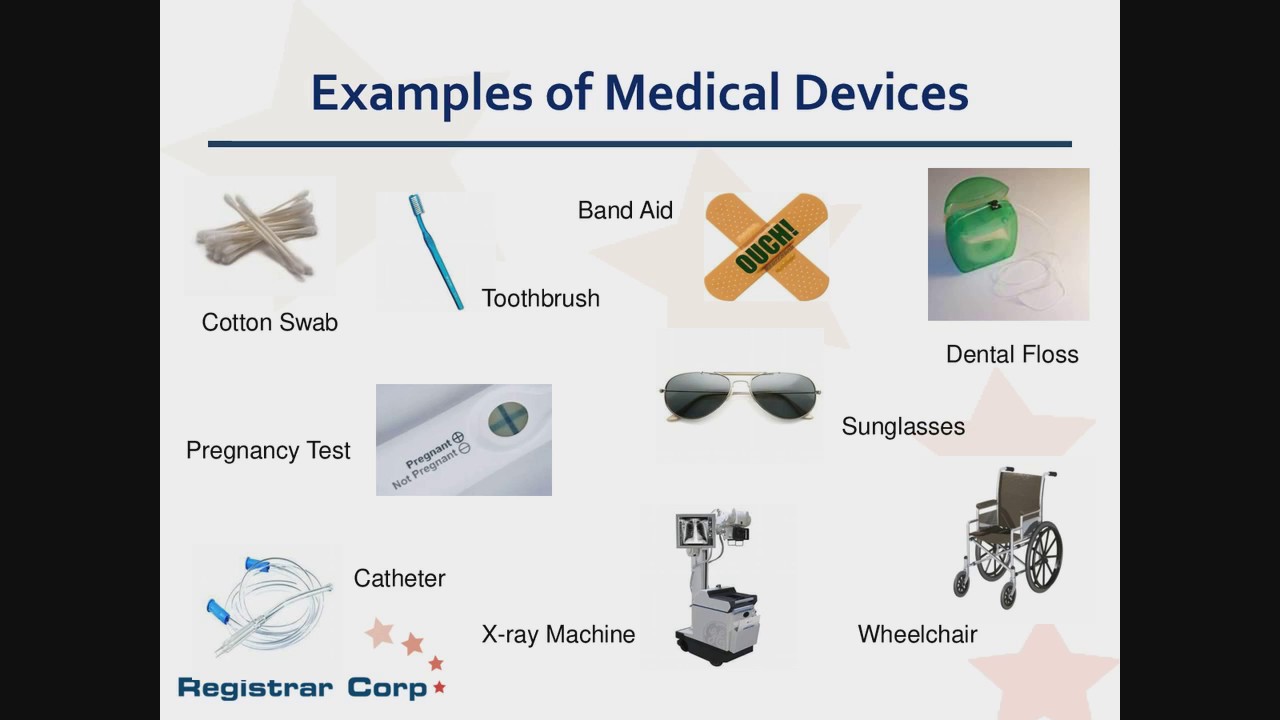
What is the fda definition of a medical device. An instrument apparatus implement machine contrivance implant in vitro. Decorative contact lenses are considered medical devices. The FDA oversees their safety and effectiveness just like contact lenses that correct your vision.
A predicate device is a medical device that may be legally marketed in the US. Lets review for each of these countries the definition of What is a medical device. The total finished design output consists of the device its packaging and labeling and the device master record.
A The MedWatch Medical Device Reporting Code Instruction Manual contains adverse event codes for use with Form FDA 3500A. FDC Act defines a device as. By Jon Speer April 7 2016 in Design Controls and User Needs.
Food and Drug Administration FDA design controls they often place a lot of emphasis on design inputs and outputs verification transfer and the design history file. In Brazil ANVISA is regulating the healthcare industry and finally in China its SFDA or CFDA. The FDA approval process for vaccines therapeutics and other pharmaceuticals was on the nightly news through late 2020.
Will be regulated by the Food and Drug Administration FDA as a medical device and is subject to premarketing and postmarketing regulatory controls. The Food and Drug Administration FDA has very specific criteria to define a medical device. Definition of a Medical Device.
This is how they determine if your product is indeed a medical device. 1 An instrument apparatus implement machine contrivance implant in vitro reagent or other similar or related article including a component part or accessory which is. Medical Device Definition an instrument apparatus implement machine contrivance implant in vitro reagent or other similar or related article including a component part or.
You may obtain the coding manual from FDAs website at. The 21 st Century Cures Act amended the language of the Federal Food Drug and Cosmetic Act FDC Act to remove said software functions from the definition of a medical device. So you will then understand What is Medical Devices Regulation In USA its FDA who is the health authority and who defines what is a medical device.
Section 201h of the Food Drug Cosmetic Act. And used as a point of comparison for new medical devices seeking approval through FDAs 510 k premarket clearance pathway. The purpose of the recent FDA ruling is to ensure that the stated scope of the agencys regulatory authority corresponds to what is currently delineated in the FDC Act.
8 Questions That Define Your Medical Device User Needs. W hen people talk about US. The new device must be proven to be substantially equivalent in safety and efficacy to the predicate device in order to receive clearance.
H Five-day report means a medical device report that must be submitted by a manufacturer to us under 80353 within 5 work days. Software that is connected to a hardware medical device but is not needed by that hardware medical device to achieve its intended medical purpose is Software as a Medical Device and not an.

Software As Medical Device Samd Classification And Definitions

Medical Devices Classification As Per Fda Medical Device Regulations Medicaldevices Fda Youtube
Https Www Fda Gov Media 131268 Download

Fda 101 For Medical Devices Youtube
