Is located in Irvine CA United States and is part of the Medical Equipment Supplies Manufacturing Industry. About Us Capillary Biomedical Capillary Biomedical Inc.
Biomedical Devices Llc Linkedin
Request more information on FDA and CE regulatory filings.
Biomedical devices irvine ca. Apply to Product Development Engineer Senior RD Engineer RD Engineer and more. Sales figure is modelled. Irvine CA 92612 3.
Is a company based out of 17171 Daimler St Irvine California United States. Full-time temporary and part-time jobs. Device Generic Name Cardiac Radio Frequency RF Ablation System Device Trade Name IBI TherapyrM Cardiac Ablation System Applicants Name Irvine Biomedical Inc.
NXT Biomedical incubator plans to spend 25 million over the next five years on cutting-edge technologies designed to address unmet needs with another 250 million pledged for creation of five to eight startups to be spun out of the incubator. Has 37 total employees across all of its locations and generates 1025 million in sales USD. Each startup will bring to market clinically relevant medical technology carefully designed to improve outcomes and reduce cost to the healthcare.
US-China Biomedical Technology Inc. Irvine CA 92614 Date of Panel None Recommendation. Company profile page for BIO-Medical Devices International Inc including stock price company news press releases executives board members and contact information.
CapBio headquartered in Irvine California is developing a novel 7-day infusion set for use with insulin pumps for the intensive management of diabetes. Premarket Approval P040014. Application PMA Date Notice of Approval of January 14 2005 Application.
Utilizing our autologous cell therapy platform which has. Free fast and easy way find a job of 711000 postings in Irvine CA and other big cities in USA. Ophthalmic Device Regulatory Testing.
We found 653 results for Medical Device Companies in or near Irvine CA. BIO-MEDICAL DEVICES INTERNATIONAL INC. With its full state-of-the-art lab in Irvine CA InVision Biomedical provides testing and technical strategy services for the submission of FDA IDE 510k and PMA and CE regulatory filings.
Bio-Med Devices has been making transport ventilators since the early 1970s. The businesses listed also serve surrounding cities and neighborhoods including Irvine CA Orange CA and Santa Ana CA. Integrated helmet design eliminates the cumbersome air tube and bulky blower unit of conventional PAPR designs and minimizes the number of accessories and peripherals needed for cost effective use on an ongoing basis.
Full-time temporary and part-time jobs. FDA-Registered Company Profile for BIO-MEDICAL DEVICES INC. University of California at Irvine Irvine CA Guann-Pynn Li Paper No.
20 Indication for Use. Apply to Intern Biomedical Technician Algorithm Engineer and more. Zip Postal Code.
They also appear in other related business categories including Hearing Aids Assistive Devices Surgical Instruments and Audiologists. Search and apply for the latest Biomedical field engineer jobs in Irvine CA. IBI and Address 2375 Morse Ave.
Free fast and easy way find a job of 758000 postings in Irvine CA and other big cities in USA. Biomedical Companies in Irvine CA 1. The CapBio team has extensive experience developing biomedical devices including insulin pumps and glucose sensors.
Apply to Assembler and more. AIVITA Biomedical is a biotechnology company developing personalized vaccines for the prevention of COVID-19 and treatment of cancer. The TV-100 offers complete patient coverage from the smallest premature infant to the largest adult and offers the latest modes graphics and waveforms a built-in turbine compressor and a 7 hour battery life.
Each team member has venture-backed startup experience and has developed multiple commercial devices. Search and apply for the latest Biomedical sciences jobs in Irvine CA. We take a unique pan-antigenic approach for our cancer vaccine targeting all neoantigens rather than a select few allowing our treatment to overcome cancers tendency to mutate over time.
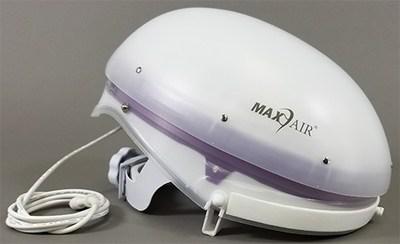
Maxair Systems Ramps Up Production To Meet Covid 19 Respiratory Protection Needs

Medical Device Regulatory Testing And Certification Ul
Https Www Flandersinvestmentandtrade Com Export Sites Trade Files Market Studies 592120917231545 592120917231545 1 Pdf

