Medical device as any product that does not achieve its purposes by chemical action or metabolization. Definition of a Medical Device.

What Is A Medical Device Official Definition For Eu Usa China Brazil
Devices in this category are capable of posing.

Fda class 2 medical device definition. Section 201h of the Food Drug Cosmetic Act. They are also considered to be at slightly higher risk than Class I devices and therefore require more stringent regulatory controls to provide assurance of their effectiveness and safety. 1 medical device means any instrument apparatus appliance software implant reagent material or other article intended by the manufacturer to be used alone or in combination for human beings for one or more of the following specific medical purposes.
As simple as a tongue depressor As complex as. Examples of Class II devices include. Condoms pregnancy testing kits and powered wheelchairs.
Class I Class II and Class III. Categorized as a Class II device for patient monitoring the PhysIQ PPA software is the first FDA-cleared device to receive the new product code PLB. Examples of devices in this category include wheel chairs X-ray machines and cardiac monitors.
Class II Devices. An instrument apparatus implement machine contrivance implant in vitro. 2 Class II means the class of devices that is or eventually will be subject to special controls.
Class I includes devices with the lowest risk and Class III includes those with. The FDA categorizes medical devices into one of three classes Class I II or III based on their risks and the regulatory controls necessary to provide a reasonable assurance of safety and. Currently medical devices fall into one of three medical device classifications.
Class III Medical Devices - devices in this class have an intricate design and support or sustain human lives. Class II devices are simple devices though they are more complicated than Class I devices. A device is in class II if general controls alone are insufficient to provide reasonable assurance of its safety and effectiveness and there is sufficient information to establish special controls including the promulgation of performance standards postmarket surveillance patient registries development and.
Devices classified into class II are devices for which special controls combined with general controls are necessary to provide reasonable assurance of safety and effectiveness. FDC Act defines a device as. Automated calculation of a summary index based on.
The class to which your device is assigned determines among other things the type of premarketing submissionapplication required for FDA clearance to market. Class II Medical Devices - devices in this class have a more complex design and pose minimal risk of injury or illness to patients or operators.

Medical Devices Classification As Per Fda Medical Device Regulations Medicaldevices Fda Youtube

Are You Making A Medical Device Voler Systems
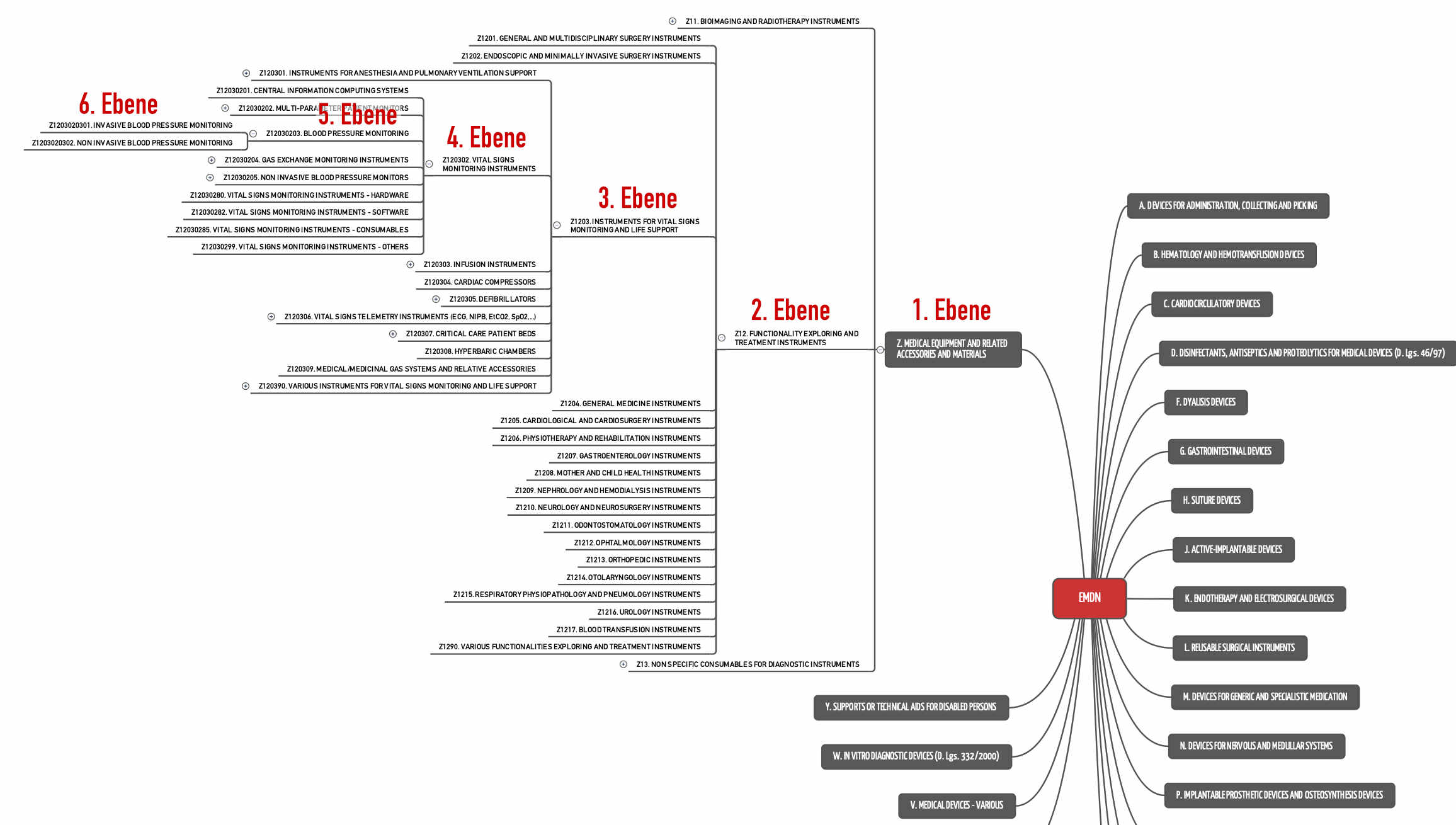
Device Group Device Category Device Group Versus Device Category

Fda Medical Device Classification Medical Device Validationpresentationeze
